In the deterministic mode MesoRD will do what we call a
mean-field approximation. The approximation is that the state,
i.e. number of molecules, change
continuously and with the average rate given by the stochastic
description at each point in time. To see what this means,
consider the events that changes the number of A-molecules in
subvolume
 during the next time period
during the next time period
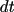 , assuming that the systems has only one spatial dimension. The
different reactions occurs with probability
, assuming that the systems has only one spatial dimension. The
different reactions occurs with probability
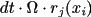 and when reaction
and when reaction
 occurs the number of A-molecules changes by
occurs the number of A-molecules changes by
 . The A-molecules can also diffuse away with probability
. The A-molecules can also diffuse away with probability
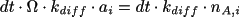 to each of the two neighbouring subvolumes in one dimension. The
number of A-molecules in subvolume
to each of the two neighbouring subvolumes in one dimension. The
number of A-molecules in subvolume
 can also increase if A-molecules diffuse from the neighbouring
subvolumes. They do this with probability
can also increase if A-molecules diffuse from the neighbouring
subvolumes. They do this with probability
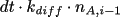 and
and
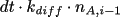 , respectively. The average change in the number of A-molecules in
subvolume
, respectively. The average change in the number of A-molecules in
subvolume
 during
during
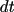 is therefore
is therefore
where the values in parenthesis are the stoichiometries for
changes of the number of A-molecules of the different
events. Similar expressions simultaneously govern the A-molecules
in other subvolumes and other species.
If we divide Equation 6.4 by
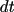 we get a system of ordinary differential
equations, for instance for
we get a system of ordinary differential
equations, for instance for
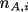
where
 . In the deterministic mode MesoRD solve this kind of ODE systems
numerically.
. In the deterministic mode MesoRD solve this kind of ODE systems
numerically.
If we divide Equation 6.5 by
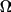 and take the limit
and take the limit
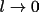 we get a more familiar expression for the reactions diffusion equation
we get a more familiar expression for the reactions diffusion equation
with
 as the continuous spatial coordinate.
as the continuous spatial coordinate.


